|
Alchemy is often associated with the quest to turn lead or other base metals into gold, though alchemists were also interested in many of the questions of modern chemistry. The word chemistry comes from a modification during the Renaissance of the word alchemy, which referred to an earlier set of practices that encompassed elements of chemistry, metallurgy, philosophy, astrology, astronomy, mysticism, and medicine. The applications of various fields of chemistry are used frequently for economic purposes in the chemical industry. It has evolved, and now chemistry encompasses various areas of specialisation, or subdisciplines, that continue to increase in number and interrelate to create further interdisciplinary fields of study. For example, chemistry explains aspects of plant growth ( botany), the formation of igneous rocks ( geology), how atmospheric ozone is formed and how environmental pollutants are degraded ( ecology), the properties of the soil on the moon ( cosmochemistry), how medications work ( pharmacology), and how to collect DNA evidence at a crime scene ( forensics).Ĭhemistry has existed under various names since ancient times. It is sometimes called the central science because it provides a foundation for understanding both basic and applied scientific disciplines at a fundamental level. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. Chemistry also addresses the nature of chemical bonds in chemical compounds. It is a physical science within the natural sciences that studies the chemical elements that make up matter and compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during reactions with other substances. Converting between Mass and Number of MolesĪ substance’s molar mass can be used to convert between the mass of the substance and the number of moles in that substance.Chemistry is the scientific study of the properties and behavior of matter. 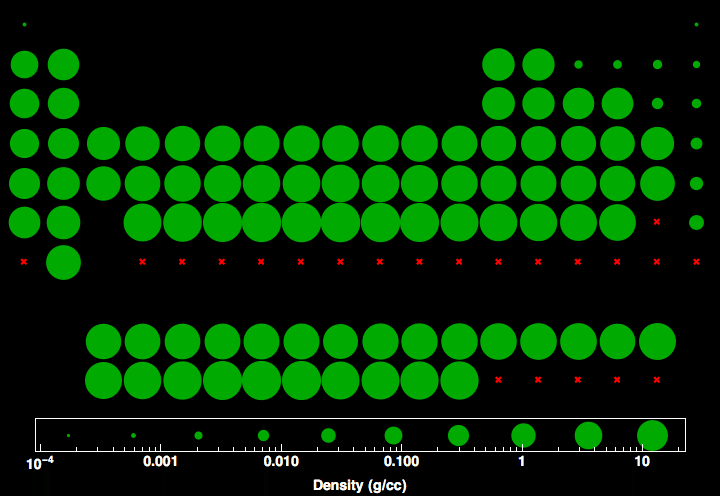
Molar Mass Calculations – YouTube: This video shows how to calculate the molar mass for several compounds using their chemical formulas. For example, since one atom of oxygen will combine with two atoms of hydrogen to create one molecule of water (\text is 58.44 g/mol. Chemical Computations with Avogadro’s Number and the MoleĪvogadro’s number is fundamental to understanding both the makeup of molecules and their interactions and combinations. So, 1 mol contains 6.022×10 23 elementary entities of the substance. It is defined as the amount of a substance that contains as many particles as there are atoms in 12 grams of pure carbon-12. The mole (abbreviated mol) is the SI measure of quantity of a “chemical entity,” such as atoms, electrons, or protons. With Avogadro’s number, scientists can discuss and compare very large numbers, which is useful because substances in everyday quantities contain very large numbers of atoms and molecules. Avogadro’s number is 6.022×10 23 molecules. It is equal to 6.022×10 23 mol -1 and is expressed as the symbol N A.Īvogadro’s number is a similar concept to that of a dozen or a gross. Avogadro’s number is defined as the number of elementary particles (molecules, atoms, compounds, etc.) per mole of a substance. Although he did not determine the exact proportion, he is credited for the idea.Īvogadro’s number is a proportion that relates molar mass on an atomic scale to physical mass on a human scale. Avogadro’s Number Amedeo Avogadro: Amedeo Avogadro is credited with the idea that the number of entities (usually atoms or molecules) in a substance is proportional to its physical mass.Īmadeo Avogadro first proposed that the volume of a gas at a given pressure and temperature is proportional to the number of atoms or molecules, The solution is the concept of the mole, which is very important in quantitative chemistry. They also need a way to compare these numbers and relate them to the weights of the substances, which they can measure and observe. It is impractical to try to count or visualize all these atoms, but scientists need some way to refer to the entire quantity. The chemical changes observed in any reaction involve the rearrangement of billions of atoms. mole: The amount of substance of a system that contains as many elementary entities as there are atoms in 12 g of carbon-12. 
For example, the mean molecular weight of water is 18.015 atomic mass units (amu), so one mole of water weight 18.015 grams.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
